From Proteins to Body Surface ECG: Computational Investigations of Ion Channelopathies
- contact:
- project group:
Cardiac modeling
From Proteins to Body Surface ECG: Computational Investigations of Ion Channelopathies
Mutations that impact on cardiac electrophysiology have been of scientific interest throughout the last decade. By altering specific ion channel characteristics, these mutations are associated with a high risk for the development of ventricular arrhythmias and sudden death. Although DNA analysis has enhanced our understanding of the underlying processes, inter-individual differences impede to quantitatively predict the effects of the mutated channels on the ECG.
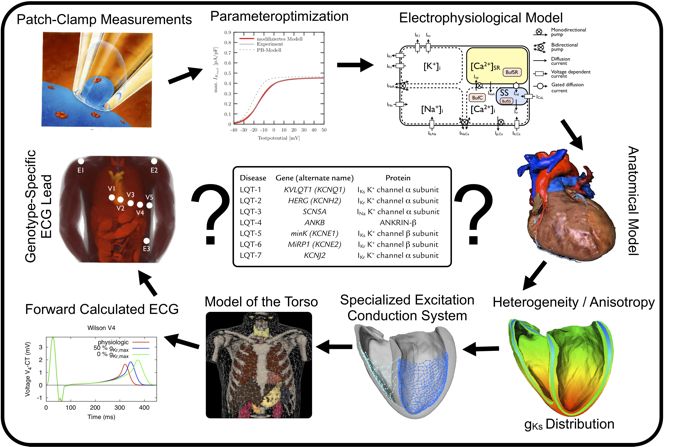
This project is focused on identifying and discriminating genotypes of mutations in a cardiac channelopathy called Long QT syndrome (LQTS) using a computational approach. Geometrical datasets of affected patients are constructed based on MRI. Together with detailed
mathematical models of cardiac electrophysiology, the excitation conduction on the individual cardiac anatomy is simulated. To this end, a patient-specific endocardial stimulation profile is created that mimicks the role of the excitation conduction system and enables a realistic depolarisation sequence. A subsequent forward calculation allows the determination of electrical signals on the body surface and the extraction of standard ECG leads. For verification and optimization of the computational model, this calculated signal on the body surface is compared to 64 channel ECG recordings from patients. Effects of the sympathetic nervous system are included into the computer model of the LQTS as heart rate and adrenergic regulation influence ion channel characteristics and thereby cardiac activity. As cardiac repolarization depends crucially on heterogeneous ion channel distributions they are integrated into the electrophysiological model as well. Especially the differences of the T wave morphology between the genotypes of LQTS will be used to derive ECG electrode positions that allow a new ECG-based discrimination of the mutation genotype.
Publications
- D.U.J. Keller et al. In-silico Evaluation of Beta-Adrenergic Effects on the Long-QT Syndrome. Proceedings Computing in Cardiology, vol. 37, pp. 825-828, 2010
- D.U.J. Keller et al. Influence of IKs Heterogeneities on the Genesis of the T-Wave: A Computational Evaluation. IEEE TBME (submitted), 2011

